The accurate clinical stratification of steatotic liver disease (SLD) remains a formidable challenge in modern hepatology, heavily confounded by the well-documented phenomenon of patient underreporting regarding alcohol consumption. Historically, differentiating pure metabolic dysfunction-associated steatotic liver disease (MASLD) from alcohol-associated liver disease (ALD) or the dual-etiology metabolic dysfunction and alcohol-associated liver disease (MetALD) has relied on patient history and direct alcohol biomarkers. However, previous prospective cohort investigations have demonstrated that approximately 16% of individuals with SLD systematically underreport their alcohol intake. Consequently, reliance on self-reporting leads to substantial clinical misclassification: in prior analyses, 16% of heavy drinkers were erroneously classified as having MASLD, while 29% were misclassified as having MetALD.
While direct measurement of whole blood phosphatidylethanol (PEth) levels serves as a highly specific and sensitive gold standard for detecting recent significant alcohol consumption, the assay is cost-prohibitive, technically demanding, and largely impractical for population-level screening or routine primary care implementation. Addressing this critical diagnostic gap, a recent study published in Gastroenterology details the development and validation of a highly scalable, cost-effective algorithmic tool named the MetALD-ALD Prediction Index (MAPI).
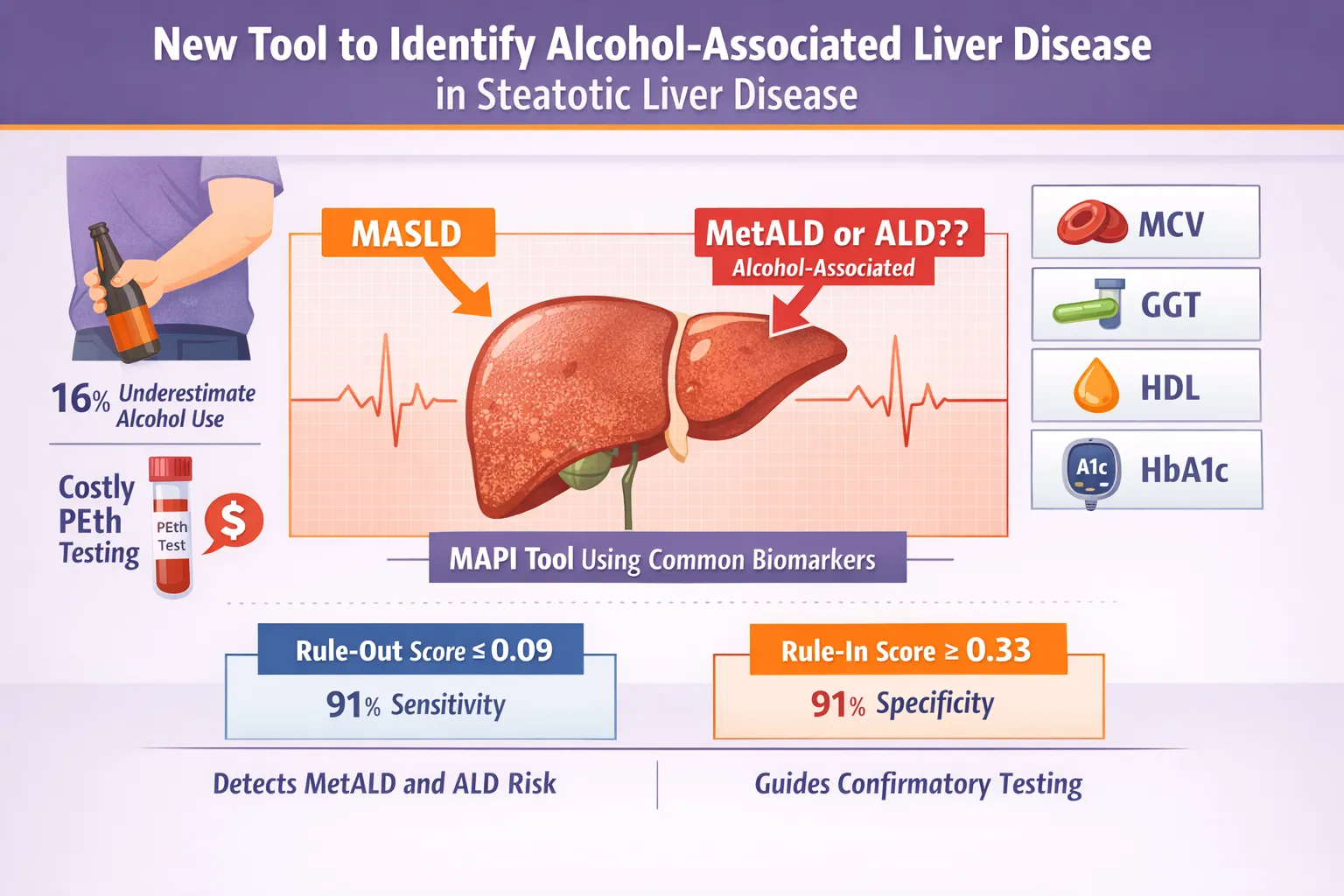
The MetALD-ALD Prediction Index (MAPI)
“Identifying alcohol-related vs. metabolic or other causes of liver disease allows clinicians to tailor management strategies, provide targeted counseling and improve patient outcomes through personalized care,” stated Federica Tavaglione, MD, PhD, a postdoctoral researcher at the University of California San Diego.
To construct a pragmatic clinical tool, Tavaglione and colleagues engineered the MAPI utilizing a composite of five routinely assessed, widely available laboratory and demographic parameters: patient sex, mean corpuscular volume (MCV), gamma-glutamyl transferase (GGT), high-density lipoprotein (HDL) cholesterol, and hemoglobin A1c (HbA1c). By integrating these variables into a predictive model, the MAPI yields a continuous probability score ranging from 0 to 1, where a score of 1 indicates a 100% probability that the patient’s hepatic steatosis is driven by significant alcohol use (MetALD or ALD).
Study Methodology and Cohort Characteristics
The investigators utilized a rigorous dual-cohort methodology for model derivation and external validation. The derivation cohort was drawn from the San Diego Liver Study, comprising 503 participants presenting with SLD and concurrent overweight or obesity. The external validation cohort consisted of a significantly larger, independent Swedish cohort of 1,777 adults who had at least one documented PEth score between July 2012 and October 2020.
Table 1. Demographic and Clinical Characteristics of the Derivation and Validation Cohorts
| Characteristic | Derivation Cohort (San Diego Liver Study) | External Validation Cohort (Swedish Cohort) |
| Total Participants (N) | 503 | 1,777 |
| Mean Age (Years ± SD) | 51 ± 13 | 57 ± 15 |
| Sex Distribution | 53% Female | 57% Male |
| Mean BMI (kg/m²) | 32.9 | 31.2 |
| Disease Classification* | ||
| MASLD | 81% | 65% |
| MetALD | 12% | 18% |
| ALD | 7% | 17% |
*Disease classification in both cohorts was strictly adjudicated utilizing a combination of self-reported alcohol consumption and objective, confirmatory PEth testing.
Diagnostic Performance and Clinical Cutoffs
The MAPI demonstrated robust discriminative capability, outperforming any single isolated biomarker (such as GGT or MCV alone) in identifying unrecognized alcohol-associated hepatic injury. In the primary San Diego derivation cohort, the MAPI achieved an overall diagnostic accuracy, defined by the area under the receiver operating characteristic curve (AUROC), of 76% (95% CI, 0.71-0.82). This performance remained highly consistent when applied to the geographically and demographically distinct Swedish validation cohort, yielding an AUROC of 75% (95% CI, 0.73-0.78).
To maximize clinical utility, the researchers established two distinct operational thresholds for the MAPI score: a highly sensitive “rule-out” cutoff and a highly specific “rule-in” cutoff.
Table 2. Diagnostic Performance of MAPI Operational Cutoffs (Derivation Cohort)
| Clinical Application | MAPI Score Threshold | Sensitivity | Specificity | Positive Predictive Value (PPV) | Negative Predictive Value (NPV) |
| Rule-Out Cutoff | $\le$ 0.09 | 91% | — | — | 94% |
| Rule-In Cutoff | $\ge$ 0.33 | — | 91% | 53% | — |
| Overall Model | Continuous (0 to 1) | 60% | 80% | — | — |
The dual-threshold approach provides clinicians with a tiered triage system. A MAPI score of $\le$ 0.09 effectively rules out clinically significant alcohol contribution with a 94% negative predictive value, confidently sparing the patient from expensive PEth testing. Conversely, a score of $\ge$ 0.33 signals a high likelihood of alcohol involvement, triggering targeted intervention or confirmatory testing.
Reflecting on the successful translation of these findings into a practical algorithm, Dr. Tavaglione remarked, “Our goal was to develop an accurate and scalable biomarker panel for MetALD and ALD, and the results — supported by independent validation — demonstrate that we achieved this objective in a clinically meaningful way.”
Ultimately, the MAPI tool acts as a critical prescreening filter in hepatology and general practice. It bridges the gap between unreliable patient self-reporting and the high costs of mass direct-biomarker screening. “These patients are strong candidates for confirmatory PEth testing, when available, enabling more informed clinical decision-making,” Tavaglione concluded, highlighting how algorithmic medicine can synthesize routine metabolic panels into powerful diagnostic insights.Show me the visualisation
Source link

