Phase 3 VALOR Trial: Oral Brepocitinib Demonstrates Significant Efficacy in Treating Dermatomyositis
Background and Clinical Context
The therapeutic landscape for dermatomyositis, a debilitating autoimmune condition characterized by progressive muscle weakness and severe cutaneous manifestations, remains profoundly constrained. As Ruth Ann Vleugels, MD, MPH, MBA, vice chair for academic affairs at Brigham and Women’s Hospital, highlighted, the standard of care relies heavily on agents with substantial toxicity profiles, and long-term remission is rare: “Most clinicians use high doses of systemic corticosteroids for essentially all patients with this disease. These drugs have substantial side effects, including bone loss, eye problems, diabetes and high blood pressure. There are no targeted modern therapies that are currently approved for dermatomyositis.”
Brepocitinib, developed by Priovant Therapeutics, represents a novel therapeutic approach as a first-in-class, oral dual inhibitor of tyrosine kinase 2 (TYK-2) and Janus kinase 1 (JAK-1). This dual-inhibition mechanism is designed to directly target the pathogenic cytokine pathways driving the disease. “This agent was studied in dermatomyositis because there is a mechanistic rationale of why it should work,” Dr. Vleugels explained. “In particular, it suppresses interferon-1, interferon-2 and several additional cytokines that are all involved in the pathogenesis of dermatomyositis.”
Study Design and Cohort Characteristics
The phase 3 VALOR trial investigated the safety and efficacy of brepocitinib in 241 adults diagnosed with definite or probable dermatomyositis. Participants were randomized in a 1:1:1 ratio to receive once-daily oral doses of brepocitinib 15 mg, brepocitinib 30 mg, or a matched placebo. The primary efficacy endpoint was the mean Total Improvement Score (TIS) at week 52, comparing the 30 mg brepocitinib arm to placebo. A critical component of the trial design involved a protocol-mandated tapering of background systemic corticosteroids between weeks 12 and 36 to a target of <5 mg per day.
Dr. Vleugels emphasized the severe, treatment-refractory nature of the cohort: “Can you imagine how challenging the state of affairs is in the therapeutic world for this disease if patients are willing to enter a yearlong placebo-controlled study? Most patients in this study were on background systemic corticosteroids; two-thirds were on two or more agents for their dermatomyositis.”
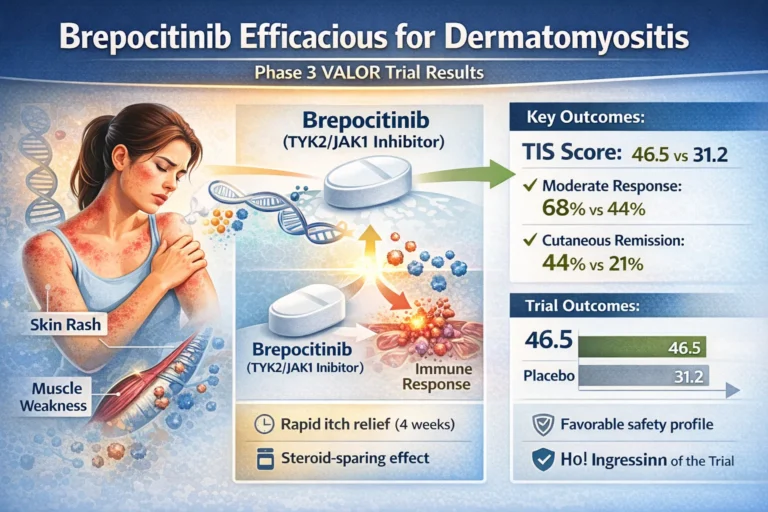
Table 1: Baseline Characteristics of the VALOR Trial Cohort
| Characteristic | Value |
| Total Participants (n) | 241 |
| Randomization Arms | Brepocitinib 15 mg (n=81); Brepocitinib 30 mg (n=81); Placebo (n=79) |
| Mean Age | 51 years |
| Sex (Female) | 77.6% |
| Background Corticosteroid Use | 75.6% (across all arms) |
Primary and Secondary Efficacy Outcomes
The findings, presented at the American Academy of Dermatology Annual Meeting and simultaneously published in The New England Journal of Medicine, demonstrated that brepocitinib 30 mg met its primary endpoint and all 10 ranked secondary endpoints. At week 52, the mean TIS score was significantly higher in the 30 mg treatment arm compared to placebo (46.5 vs. 31.2; P < .001).
Table 2: Efficacy Outcomes at Week 52 (Brepocitinib 30 mg vs. Placebo)
| Clinical Endpoint | Brepocitinib 30 mg | Placebo | Statistical Difference | P Value |
| Mean Total Improvement Score (TIS) | 46.5 | 31.2 | Least-squares mean diff: 15.3 (95% CI, 6.7-24) | < .001 |
| Moderate Response (TIS $\ge$ 40) | 68% | 44% | Difference: 24% (95% CI, 7.1-37.3) | .004 |
| Major Response (TIS $\ge$ 60) | 46% | 26% | Difference: 20% (95% CI, 4.2-34.8) | .01 |
| Cutaneous Remission (CDASI $\le$ 5) | 44% | 21% | Difference: 23% | .006 |
Beyond composite scores, the drug provided substantial dermatologic and symptomatic relief. Dr. Vleugels noted the rapid onset of action regarding pruritus: “Brepocitinib has an impressive impact on itch as early as 4 weeks after initiation. For dermatologists, one of the challenges they encounter when treating dermatomyositis is the intense itch that goes along with the disease, in addition to the severe rash. Thankfully, both the itch and rash responded extremely well.”
Furthermore, patients in the 30 mg arm demonstrated statistically significant improvements in proximal muscle strength, functional disability (P = .0035), and overall quality of life. Notably, these superior clinical outcomes were achieved concurrently with greater reductions in systemic corticosteroid use among the brepocitinib group compared to the placebo group.
Safety Profile
The trial enrolled a highly comorbid, “real-world” population; 20% had active interstitial lung disease, 14% had a history of malignancy, and approximately 66% possessed at least one cardiovascular risk factor.
Safety analyses revealed a numerical increase in serious infections within the brepocitinib 30 mg group, an adverse event profile consistent with the established class effects of TYK-2 and JAK-1 inhibitors. However, Dr. Vleugels clarified that these events did not broadly preclude continued treatment: “These participants were able to complete the study and remain on brepocitinib and actually have good outcomes in their myositis.”
Crucially, major safety concerns often associated with JAK inhibitors were not elevated in the active treatment arm. One cardiovascular event was observed in the 30 mg group compared to two in the placebo group. Furthermore, Dr. Vleugels stressed, “Very importantly, clots and malignancies were only seen in the placebo group.” There were zero fatalities recorded during the 52-week study period.
Conclusions and Regulatory Outlook
The robust data generated by the VALOR trial positions brepocitinib as a highly promising targeted therapy for a disease state historically characterized by treatment failure. “There have been well over a decade of failed studies in dermatomyositis,” Dr. Vleugels stated. “To have a pivotal phase 3 global randomized control trial that hit all these endpoints is exciting for the myositis community.”
The FDA granted brepocitinib priority review status in March, with a target action date anticipated in the third quarter of 2026. If approved, brepocitinib will provide clinicians with a targeted, steroid-sparing oral therapy, marking what Dr. Vleugels described as “a major move forward for our patients.”

