Zongertinib as First-Line and CNS-Active Therapy for HER2-Mutant Advanced Non-Small Cell Lung Cancer: Updated Results from the Beamion LUNG-1 Study
Conference Presentation Summary with Clinical Commentary
Heymach JV, et al. Abstract 6MO. Presented at: European Lung Cancer Congress; March 25–28, 2026; Copenhagen, Denmark.
Importance
HER2 mutations occur in approximately 3% to 4% of patients with non-small cell lung cancer (NSCLC) — a relatively small proportion that nonetheless represents a population of substantial absolute size given the overall prevalence and mortality burden of lung cancer. Until recently, no HER2-directed therapy was available in the first-line setting. Updated results from the Beamion LUNG-1 study, presented at the European Lung Cancer Congress, demonstrate that zongertinib (Hernexeos, Boehringer Ingelheim) produces response rates and progression-free survival outcomes that substantially exceed both historical chemotherapy benchmarks and prior HER2-directed agents — establishing it as a meaningful first-line advance for this molecularly defined population.
Regulatory Context
The FDA granted accelerated approval of zongertinib for previously treated patients with HER2-mutant advanced NSCLC in August 2025, based on cohort 1 data from Beamion LUNG-1 demonstrating a 71% objective response rate (ORR) and a median duration of response (DoR) of 14.1 months. The approval was subsequently expanded to the first-line setting in February 2026, based on extended results from the same study. Full approval is contingent on results from the confirmatory Beamion LUNG-2 randomized phase 3 trial.
Study Design: Beamion LUNG-1 Cohort Structure
Table 1: Beamion LUNG-1 Study Cohorts
| Cohort | Population | Prior Reporting |
|---|---|---|
| 1 | Previously treated, HER2 TKD mutations | Yes — basis for initial accelerated approval |
| 2 | Previously untreated (first-line) | Presented at ELCC 2026 |
| 3 | Non-TKD mutations | Yes |
| 4 | Active brain metastases | Presented at ELCC 2026 |
| 5 | Prior HER2-directed treatment, TKD mutations | Yes |
TKD = tyrosine kinase domain
Historical Benchmarks for Contextual Interpretation
John V. Heymach, MD, PhD, chair of thoracic and head and neck medical oncology at The University of Texas MD Anderson Cancer Center, provided the reference points against which current results should be evaluated.
Table 2: Historical Efficacy Benchmarks in HER2-Mutant NSCLC
| Treatment / Setting | ORR | Median PFS |
|---|---|---|
| Platinum doublet chemotherapy ± immunotherapy (first-line, standard of care) | ~30% | ~5–7 months |
| Trastuzumab deruxtecan (second-line) | 49% | 9.9 months |
| Poziotinib (earlier TKI; first in class with activity) | ~30% | ~5.5 months |
| Zongertinib — previously treated (cohort 1, approval basis) | 71% | Not reported here |
Dr. Heymach characterized chemotherapy with or without immunotherapy as “really the yardstick” in the first-line setting, noting expected ORRs of approximately 30% and PFS of 5 to 7 months — comparable to outcomes seen with platinum doublet therapy in EGFR-mutant NSCLC.
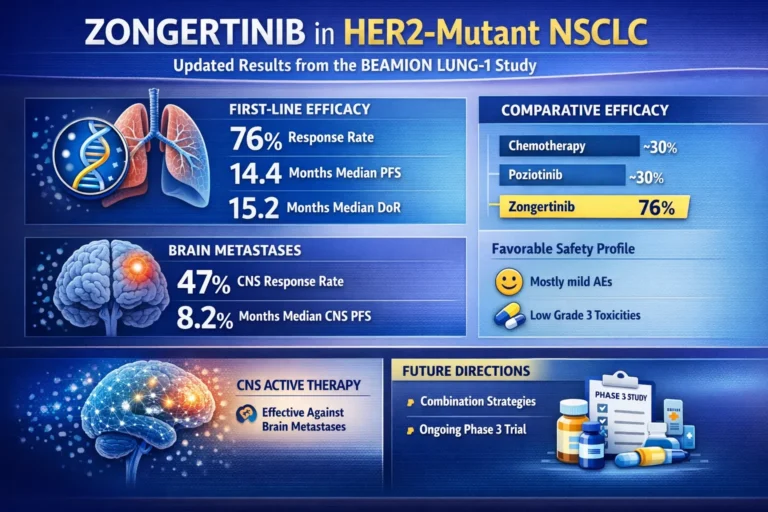
Cohort 2: First-Line Efficacy Results (n = 74)
Table 3: Cohort 2 — First-Line Zongertinib Efficacy
| Endpoint | Result | 95% CI |
|---|---|---|
| Objective response rate (ORR) | 76% | 65%–84% |
| Median duration of response (DoR) | 15.2 months | 9.8 months – not evaluable |
| Median progression-free survival (PFS) | 14.4 months | 11.1 months – not evaluable |
Dr. Heymach placed these results in direct comparative perspective:
“With earlier tyrosine kinase inhibitors that we tested — poziotinib, for example, the first tyrosine kinase inhibitor that really had substantial activity in this population — the PFS was about 5.5 months, about a 30% response rate and a lot of toxicity that went along with that. Now, roughly tripling the progression-free survival compared to our earlier tyrosine kinase inhibitors and more than doubling the response rate, we’re delighted to see that this represents a real advance for patients that exceeds, by a substantial margin, the activity that has been seen for other drugs that have been tested to date.”
He characterized the data as meeting his high expectations: “The results here are pretty clear” and called the development “the breakthrough the field has been waiting for.”
Cohort 4: Brain Metastases Results (n = 30)
Table 4: Cohort 4 — Intracranial Efficacy in Active Brain Metastases
| Endpoint | Result | 95% CI |
|---|---|---|
| Intracranial ORR (all cohort 4 patients) | 47% | 30%–64% |
| Median intracranial DoR | 6.9 months | 2.9 months – not evaluable |
| Median intracranial PFS | 8.2 months | 4.1–11.3 months |
| Intracranial ORR (confirmed measurable CNS disease, no prior brain RT) | 59% | 36%–78% |
| Median intracranial DoR (no prior brain RT subgroup) | 6.2 months | 2.7 months – not evaluable |
Dr. Heymach stated: “I think we can say zongertinib has clear evidence of CNS activity. Is there room for improvement down the road? Quite potentially. We saw this with EGFR inhibitors where, early on, we found some drugs had CNS activity, but over time, there were more and more.”
Safety Profile: Cohort 2
Table 5: Treatment-Related Adverse Events — Cohort 2
| Adverse Event | Incidence | Notes |
|---|---|---|
| Any treatment-related AE | 91% | Most mild to moderate |
| Grade ≥3 treatment-related AEs | 19% | — |
| Diarrhea (any grade) | ~50% | Cases predominantly mild; almost never required additional medication |
| Grade 3 diarrhea | 2 patients | Uncommon |
| Grade 3 rash | 0% | Zero cases |
| Rash (any grade) | Occurred | Mild and manageable |
| Liver enzyme elevation | Present | Asymptomatic |
| Paronychia | Present | Mild; manageable |
Dr. Heymach characterized the overall safety profile as “very favorable,” noting specifically that “for some drugs in this class, rash can be an issue, but there were zero patients who had grade 3 rash. When a rash did occur, it was very mild and manageable.”
Comparative Efficacy Summary
Table 6: Zongertinib vs. Historical Standards — Efficacy Comparison
| Comparator | ORR | Median PFS | Setting |
|---|---|---|---|
| Chemotherapy ± immunotherapy | ~30% | ~5–7 months | First-line (current SOC) |
| Poziotinib | ~30% | ~5.5 months | Previously treated |
| Trastuzumab deruxtecan | 49% | 9.9 months | Second-line |
| Zongertinib (cohort 1) | 71% | Not reported | Previously treated |
| Zongertinib (cohort 2) | 76% | 14.4 months | First-line |
Future Directions
Dr. Heymach framed current results as a foundation rather than a ceiling, describing zongertinib as “the starting point.” A median PFS of 14.4 months, while representing a substantial advance, falls short of the multi-year PFS benchmarks seen with targeted therapies in EGFR- and ALK-driven NSCLC — the aspirational goal for HER2-directed therapy.
Identified research priorities include combination strategies — most notably pairing zongertinib with trastuzumab deruxtecan: “I think there are some combinations that are pretty obvious, like combining our best antibody-drug conjugate, which right now is trastuzumab deruxtecan, with zongertinib.” Investigation of resistance mechanisms is also underway, with emerging data identifying mutations conferring zongertinib resistance that may be susceptible to alternative HER2-directed agents. Multiple studies are additionally examining zongertinib in HER2-mutant tumors beyond NSCLC.
The Beamion LUNG-2 randomized phase 3 trial — comparing zongertinib against platinum doublet chemotherapy with immunotherapy in previously untreated patients — is ongoing and will serve as the confirmatory study for full FDA approval.
Disclosures: Heymach reports advisory board roles with AbbVie, Amgen, AnHeart Therapeutics, Arrivent, AstraZeneca, BioNTech AG, Boehringer Ingelheim, Bristol Myers Squibb, Curio Science, DAVA Oncology, Eli Lilly, EMD Serono, Janssen, Jazz Pharmaceuticals, Mirati Therapeutics, Moffitt Cancer Center, ModeX, Novartis, OncoCyte, Pfizer, Sanofi, Spectrum Pharmaceuticals, and Takeda; and research grants from AstraZeneca, Boehringer Ingelheim, Bristol Myers Squibb, Mirati, Takeda, and Taiho. Full author disclosures available in the primary abstract.