Evolocumab in High-Risk Diabetic Patients Without Established Atherosclerosis: A Subgroup Analysis of the VESALIUS-CV Trial
Background and Rationale
The management of cardiovascular (CV) risk in patients with diabetes mellitus has historically been stratified by the presence or absence of established atherosclerotic cardiovascular disease (ASCVD). While the benefit of intensive lipid-lowering therapy, particularly with proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors, is well-documented in secondary prevention, its role in primary prevention for high-risk diabetic cohorts remains an area of active investigation. The VESALIUS-CV trial previously demonstrated that evolocumab (Repatha, Amgen) effectively reduced major adverse cardiovascular events (MACE) in a broad population of high-risk patients with diabetes and/or atherosclerosis but no history of myocardial infarction (MI) or stroke. To further elucidate the preventive capabilities of PCSK9 inhibition, a pre-specified subgroup analysis focusing exclusively on diabetic patients without known atherosclerosis was conducted. These findings were presented at the American College of Cardiology (ACC) Scientific Session in New Orleans and published concurrently in JAMA.
Study Design and Patient Population
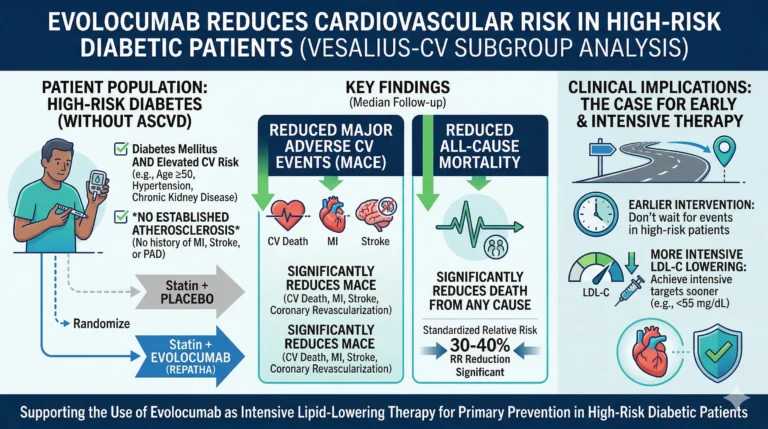
The subgroup analysis focused on 3,655 high-risk patients with diabetes who had no clinical or radiographic evidence of significant atherosclerosis. This cohort represented approximately 30% of the total VESALIUS-CV trial population.
The inclusion criteria strictly defined the absence of significant atherosclerosis as having no prior arterial revascularization, no arterial stenosis of 50% or greater, and a coronary artery calcium (CAC) score of less than 100 Agatston units. Furthermore, to qualify as “high-risk,” patients were required to have diabetes with at least one of the following characteristics: a disease duration of at least 10 years, daily use of insulin, or the presence of established microvascular disease.
Despite receiving stable statin therapy for a minimum of two weeks, all enrolled patients exhibited persistently elevated atherogenic lipoproteins at baseline, defined as:
-
Low-density lipoprotein cholesterol (LDL-C) $\ge$ 90 mg/dL, OR
-
Non-high-density lipoprotein cholesterol (non-HDL-C) $\ge$ 120 mg/dL, OR
-
Apolipoprotein B (ApoB) $\ge$ 80 mg/dL.
The median age of the cohort was 65 years, and 57% were women. Patients were randomized to receive either evolocumab or a matched placebo, in addition to their background statin therapy.
Table 1: Baseline Characteristics and Inclusion Criteria
| Parameter | Criteria/Value |
| Cohort Size | 3,655 patients |
| Median Age | 65 years |
| Sex | 57% Women |
| Diabetes Status | High-risk (duration $\ge$ 10 years, insulin use, or microvascular disease) |
| Atherosclerosis Status | No significant atherosclerosis (No prior revascularization, stenosis < 50%, CAC < 100) |
| Baseline Lipids (on statin) | LDL-C $\ge$ 90 mg/dL, OR non-HDL-C $\ge$ 120 mg/dL, OR ApoB $\ge$ 80 mg/dL |
Results: Lipid Lowering Efficacy
Evolocumab demonstrated profound and sustained reductions in LDL-C levels compared to placebo. At 48 weeks, the median LDL-C plummeted to 52 mg/dL in the evolocumab arm, compared to 111 mg/dL in the placebo arm. This robust treatment effect was maintained at 96 weeks, with median LDL-C levels of 44 mg/dL and 105 mg/dL in the evolocumab and placebo groups, respectively. Nicholas A. Marston, MD, MPH, a preventive cardiologist at Brigham and Women’s Hospital and the study’s lead presenter, stated during a press conference: “As expected, evolocumab dramatically lowered LDL cholesterol compared to placebo.”
Results: Cardiovascular Outcomes
Over a median follow-up period of 4.8 years, the intensive reduction of LDL-C translated into significant clinical benefits. The study evaluated two primary composite endpoints, alongside individual cardiovascular and all-cause mortality outcomes.
The first primary composite endpoint, consisting of cardiovascular death, myocardial infarction (MI), or ischemic stroke, was significantly reduced in the evolocumab group. This endpoint occurred in 5.0% of patients treated with evolocumab compared to 7.1% of patients receiving placebo (Hazard Ratio [HR] = 0.69; 95% Confidence Interval [CI], 0.52-0.91; P = .009). The absolute risk reduction between the groups was 2.1 percentage points.
The second, broader primary composite endpoint—which added arterial revascularization to the initial composite—also demonstrated a significant reduction. It occurred in 7.6% of the evolocumab group versus 10.5% of the placebo group (HR = 0.69; 95% CI, 0.55-0.86; P = .001), yielding an absolute risk reduction of 2.9 percentage points.
Furthermore, mortality outcomes favored the investigational arm. All-cause mortality occurred in 7.8% of the evolocumab group compared to 10.1% of the placebo group (HR = 0.76; 95% CI, 0.61-0.95; P = .017). Specifically, cardiovascular death was reduced, occurring in 2.6% of patients on evolocumab versus 4.0% on placebo (HR = 0.68; 95% CI, 0.46-0.99; P = .046).
Table 2: Clinical Outcomes at Median 4.8 Years Follow-Up
| Endpoint | Evolocumab (%) | Placebo (%) | Absolute Difference | Hazard Ratio (95% CI) | P-Value |
| Primary Composite 1 (CV Death, MI, Ischemic Stroke) | 5.0% | 7.1% | 2.1% | 0.69 (0.52-0.91) | .009 |
| Primary Composite 2 (CV Death, MI, Ischemic Stroke, Revascularization) | 7.6% | 10.5% | 2.9% | 0.69 (0.55-0.86) | .001 |
| All-Cause Mortality | 7.8% | 10.1% | 2.3% | 0.76 (0.61-0.95) | .017 |
| Cardiovascular Death | 2.6% | 4.0% | 1.4% | 0.68 (0.46-0.99) | .046 |
Clinical Implications and Expert Perspective
The findings from this subgroup analysis provide compelling evidence for the early intensification of lipid-lowering therapy in high-risk diabetic patients, even before the clinical manifestation of atherosclerotic disease. Dr. Marston emphasized this point, stating:
“The benefit seen in this subgroup supports intensification of lipid-lowering therapy beyond statins earlier in the atherosclerotic cardiovascular disease process.”
He further elaborated on the therapeutic targets, noting that the data “supports lowering LDL cholesterol down to 40 mg/dL in these patients. These data strongly support that in these lower-risk patients, we should be targeting LDL cholesterol goals that are typically reserved for very high-risk secondary prevention patients.”
The broader impact of these results on clinical practice guidelines was highlighted by Christopher M. Kramer, MD, President of the American College of Cardiology and Chief of the Cardiovascular Division at UVA Health. Dr. Kramer contextualized the findings within the evolving “lower is better” paradigm for LDL-C management. “These results expand our understanding of ‘lower is better’ for LDL. Patients with diabetes but without established severe coronary artery disease who had aggressive LDL lowering with a PCSK9 inhibitor had improved CV outcomes and mortality,” Kramer observed.
Dr. Kramer suggested that these data challenge current clinical thresholds, stating,
“I think this will change clinical practice. The recent lipid guidelines were finished before the main results of VESALIUS-CV were released. The goals were LDL less than 70 mg/dL for high-risk groups and less than 55 mg/dL for very high-risk groups.”
Referencing the implications of the VESALIUS-CV data, he argued that the aggressive target of < 55 mg/dL should be applied more broadly. “And now these results expand the number of groups where the goal should be lower. It does not matter how you get there: a statin alone, a statin plus another oral drug or a PCSK9 inhibitor. The importance is to get to goal at 55 mg/dL and below.” Dr. Kramer concluded by reaffirming the foundational biological principle supported by the trial: “The curve of LDL vs. events shows very consistent results. It’s a straight line: The lower the LDL goes, the lower the CV event rate.”

